
Understanding hydrothermal transformation from Mn2O3 particles to Na0.55Mn2O4·1.5H2O nanosheets, nanobelts, and single crystalline ultra-long Na4Mn9O18 nanowires – topic of research paper in Nano-technology. Download scholarly article PDF and read for ...

Evaluation of MnOx, Mn2O3, and Mn3O4 Electrodeposited Films for the Oxygen Evolution Reaction of Water | The Journal of Physical Chemistry C

Tuning Single-Atom Dopants on Manganese Oxide for Selective Electrocatalytic Cyclooctene Epoxidation | Journal of the American Chemical Society

Kinetics of Mn2O3–Mn3O4 and Mn3O4–MnO Redox Reactions Performed under Concentrated Thermal Radiative Flux | Energy & Fuels
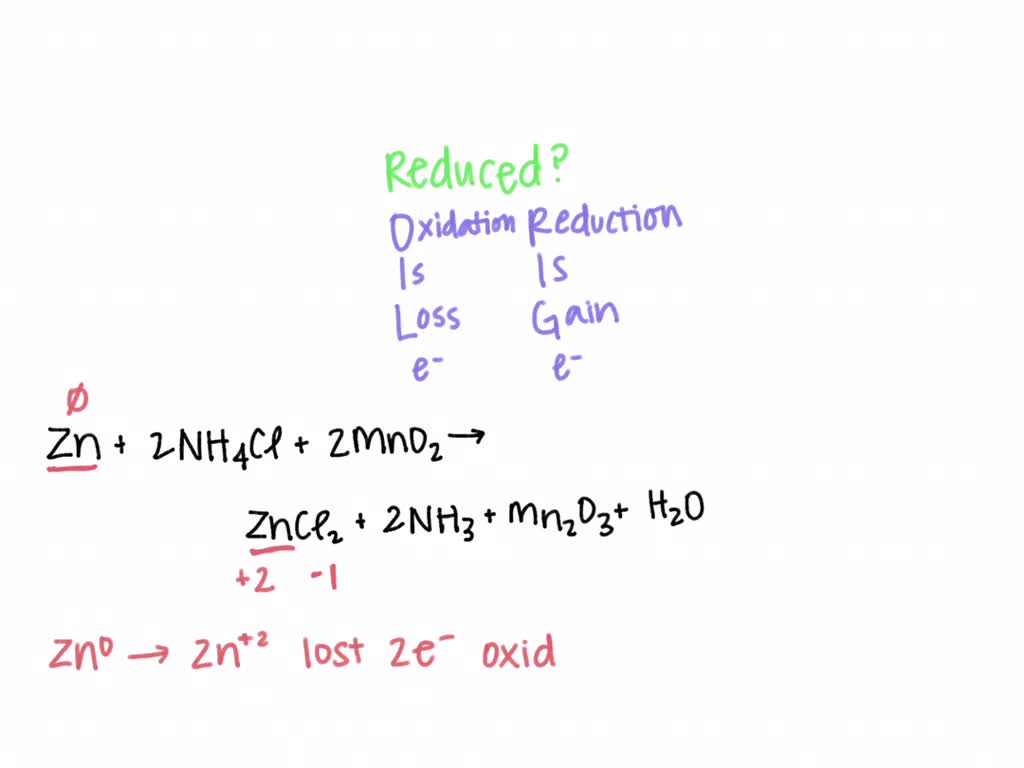
SOLVED: In an acid dry cell battery for which the reaction is Zn + 2 NH4Cl + 2 MnO2 ? ZnCl2 + 2 NH3 + Mn2O3 + H2O, what atom is reduced?

Irregularly Shaped Mn2O3 Nanostructures with High Surface Area for Water Oxidation | ACS Applied Nano Materials

XANES spectra of Mn K-edge; (a) Mn2O3, (b) Mn3O4, (c) MnO2, (d) MnO,... | Download Scientific Diagram
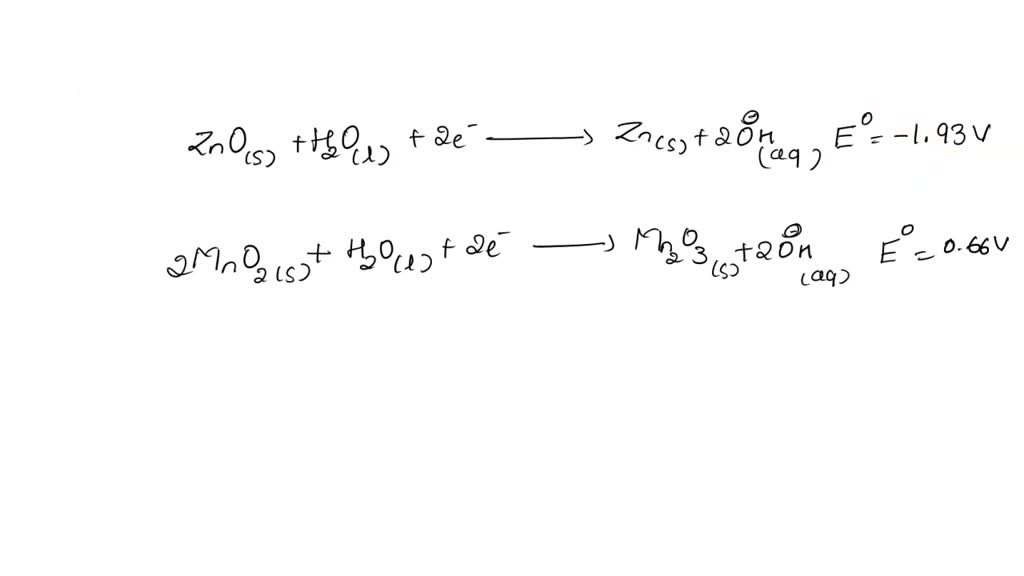
SOLVED: The standard reduction potentials of the half-reactions in single-use alkaline batteries are: ZnO(s) + H2O(l) + 2e- –> Zn(s) + 2OH-(aq) Eo = -1.93V 2MnO2(s) + H2O(l) + 2e- –> Mn2O3(s) +
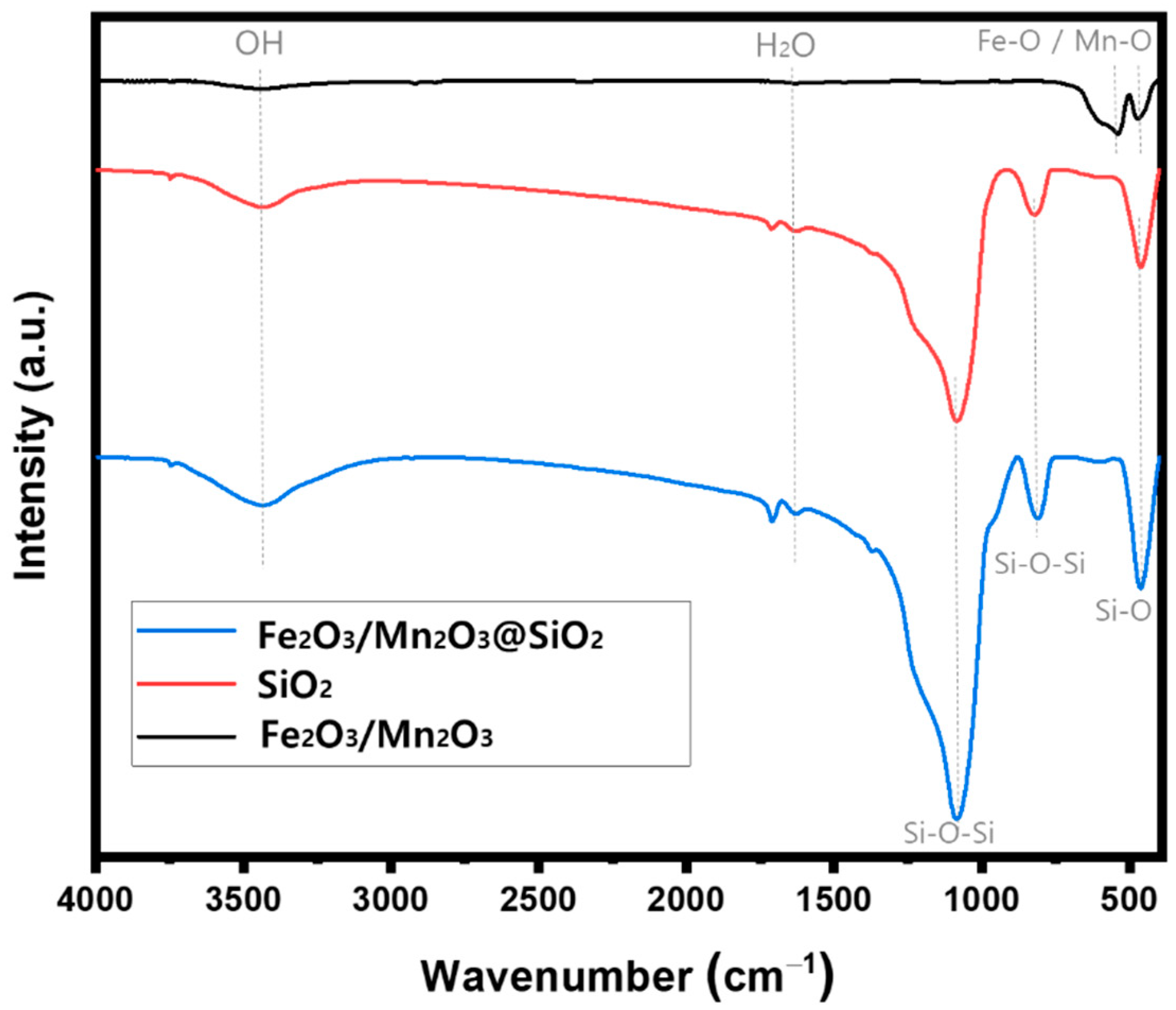
Catalysts | Free Full-Text | Synthesis of Fe2O3/Mn2O3 Nanocomposites and Impregnated Porous Silicates for Dye Removal: Insights into Treatment Mechanisms
Density functional theory calculated reaction pathways of (a) Mn2O3 +... | Download Scientific Diagram

The Most Active Oxidase‐Mimicking Mn2O3 Nanozyme for Biosensor Signal Generation - Chen - 2021 - Chemistry – A European Journal - Wiley Online Library


![Punjabi] Mn2O3 is acidic/basic oxide. Punjabi] Mn2O3 is acidic/basic oxide.](https://static.doubtnut.com/ss/web-overlay-thumb/13363282.webp)





![How many grams Mn2O3 would be produced from the complete reaction of 46.8 g of M || 102 [algebra] How many grams Mn2O3 would be produced from the complete reaction of 46.8 g of M || 102 [algebra]](https://p16-ehi-va.gauthmath.com/tos-maliva-i-ejcjvp0zxf-us/33c8cdf452324a458870f75e4227d3cb~tplv-ejcjvp0zxf-webp.webp)

