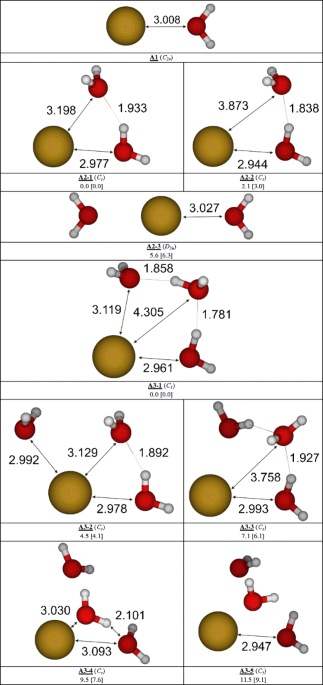
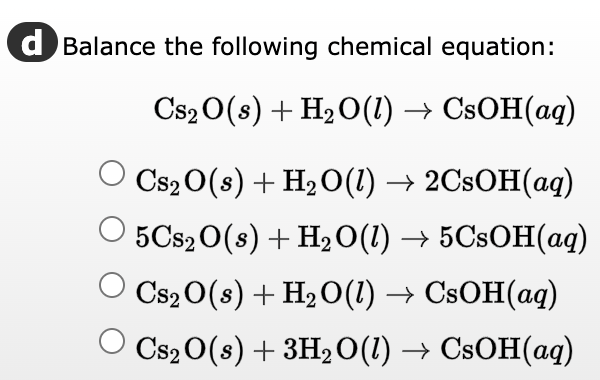Optimized structures of the H2O-Br2 + complex calculated at the UMP2... | Download Scientific Diagram

Cs + H2O = CsOH + H2 Balanced Equation||Caesium +water=Caesium hydroxide +Hydrogen Balanced Equation - YouTube

SOLVED: 11. Water gas is the name for the mixture of CO and H2 prepared by the reaction of steam with carbon at 1000°C. C(s) + H2O(g) â†' CO(g) + H2(g) ("Water

SOLVED: Consider the chemical reaction: C(s) + H2O(g) â†' CO(g) + H2(g) How many liters of hydrogen gas are formed from the complete reaction of 15.7 g C? Assume that the hydrogen
Microhydration of caesium compounds: Cs, CsOH, CsI and Cs2I2 complexes with one to three H2O molecules of nuclear safety interest | Journal of Molecular Modeling

For the water gas reaction C(s) + H2O(g) CO(g) + H2O(g) the standard Gibbs free energy of reaction (at 1000K ) is - 8.1kJ/mol . Calculate its equilibrium constant.
Isomer-specific cryogenic ion vibrational spectroscopy of the D2 tagged Cs +(HNO3)(H2O)n=0-2 complexes: Ion-driven enhancement of
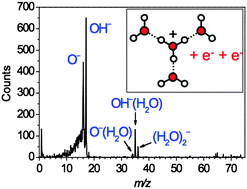
The reduction of water clusters H+(H2O)n to (OH−)(H2O)m by double electron transfer from Cs atoms - Physical Chemistry Chemical Physics (RSC Publishing)