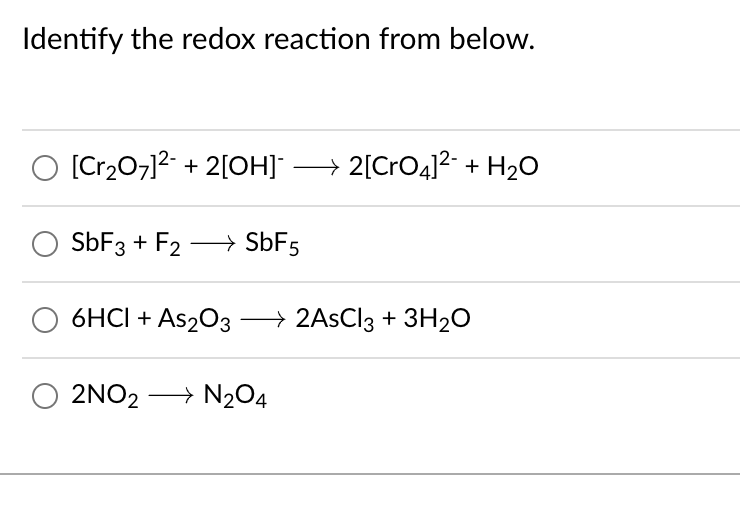Question Video: Determining the Coefficients Needed to Balance the Equation That Describes the Marsh Test | Nagwa

SOLVED: Balance the chemical equation by adding coefficients as needed. Equation: HCl(aq) + As2O3(s) + NaNO2(aq) + H2O(l) â†' NO(g) + H3AsO4(aq) + NaCl(aq)

SOLVED: A solution of iodine was standardized by titration with H3AsO3 prepared from weighing and dissolving 141.6 mg of pure As2O3. The titration reaction is H3AsO3 + I2 + H2O = H3AsO4 +

SOLVED: Balance the chemical equation by adding coefficients as needed. equation: HCl(aq) + As2O3(s) + NaNO3(aq) + H2O(l) â†' NO(g) + H3AsO4(aq) + NaCl(aq)

SOLVED: What is the stoichiometric coefficient for H2O when the following equation is balanced using the lowest, whole-number coefficients? As2O3 + H2O → H3AsO4 + H2
Może ktoś rozwiązać redoxa, robiąc to bilansem elektronowo-jonowym?As2O3 + HNO3 + H2O -> H3AsO4 + N2O3 - Brainly.pl

SOLVED: Balance the chemical equation by adding coefficients as needed. Equation: HCl(aq) + As2O3(s) + NaNO2(aq) + H2O(l) â†' NO(g) + H3AsO4(aq) + NaCl(aq)

SOLVED: 7-12. Arsenic(III) oxide (As2O3) is available in pure form and is a useful (but carcinogenic) primary standard for oxidizing agents such as MnO4. The As2O3 is dissolved in base and then
Balance the following equation using oxidation number method AS2S3 + HNO3 + H2O → H3ASO4 + H2SO4 + NO - Sarthaks eConnect | Largest Online Education Community

SOLVED: When this equation is properly balanced, the coefficients of As2O3 and H2O are, respectively, As2O3 + H2O â†' HAsO3 + ? 0l4 and ? 0l2 and ? Oland 2 Ol3ald 1