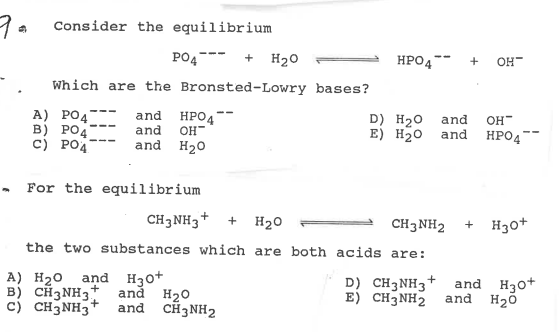
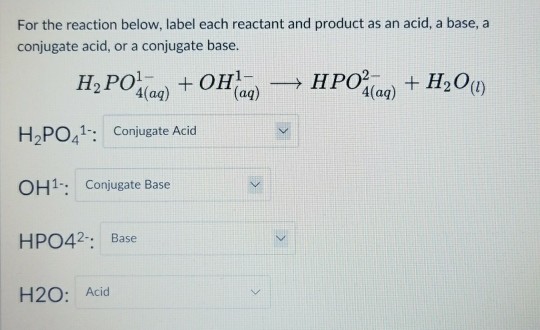
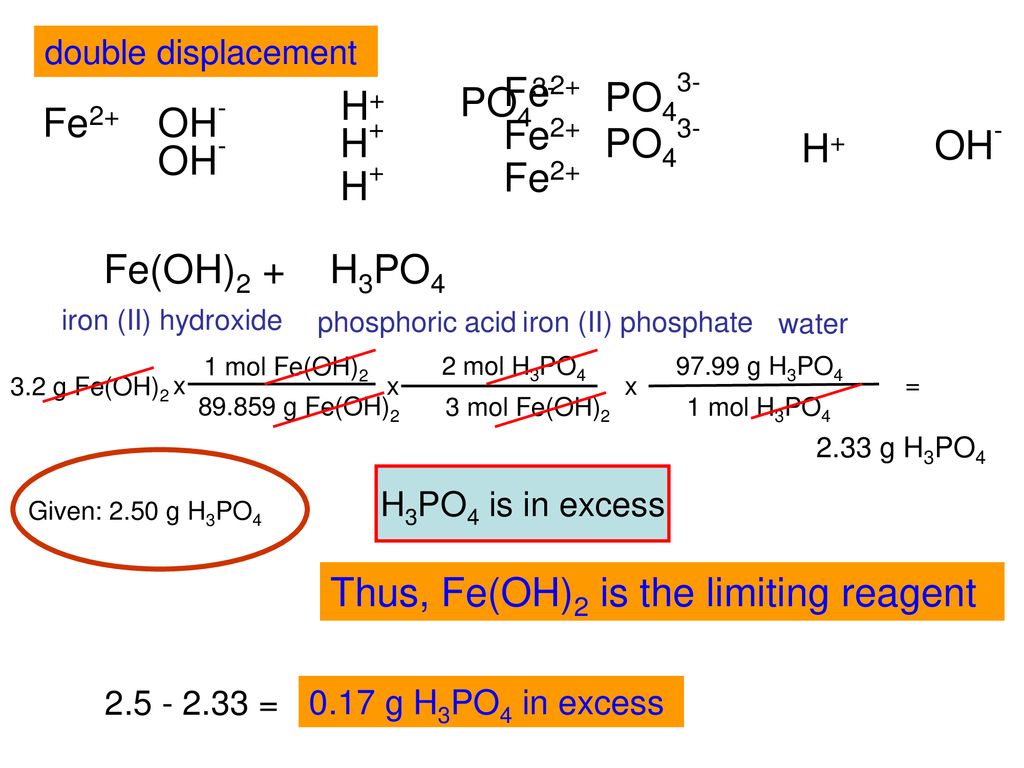
HOW TO BALANCE H3PO4+Fe(OH)2=H2O+Fe3(PO4)2 | HOW TO BALANCE H3PO4+Fe(OH)2= H2O+Fe3(PO4)2 | By Chemistry 360 | Facebook

Contribution à l'étude des phosphates ammoniaco˗métalliques monohydratés. II. Structure cristalline du phosphate de cobalt˗ammonium monohydraté : Co NH4 PO4, H2O - Persée

H3PO4+Ca(OH)2=Ca3((PO4)2+H2O balance the chemical equation @mydocumentary838. h3po4+ca(oh)2= - YouTube

SOLVED: When the equation Ca(OH)2 + H3PO4 –> Ca3(PO4)2 + H2O is balanced, the coefficient for H2O is

Synthesis and Characterization of a Novel Hydrated Layered Vanadium(III) Phosphate Phase K3V3(PO4)4·H2O: A Functional Cathode Material for Potassium-Ion Batteries | ACS Omega

SOLVED: 1. Balance the equation H3PO4 + Ca(OH)2 —> Ca3(PO4)2 + H2O and show the tally table. 2. Draw a colored particle diagram of your total balanced chemical equation. Show a key.

Figure 1 from Electrochemical analysis of Co3(PO4)2·4H2O/graphene foam composite for enhanced capacity and long cycle life hybrid asymmetric capacitors | Semantic Scholar

The experimental X-Ray diffraction pattern of Fe3(PO4)2.8H2O, allowed... | Download Scientific Diagram

La vanmeersscheite U(UO2)3(PO4)2(OH)6 • 4 H2O et la méta-vanmeersscheite U(UO2)3(PO4)2(OH)6 • 2 H2O, nouveaux minéraux - Persée

Electrochemical performances of all the prepared NH4Co x Ni1−x PO4·H2O... | Download Scientific Diagram