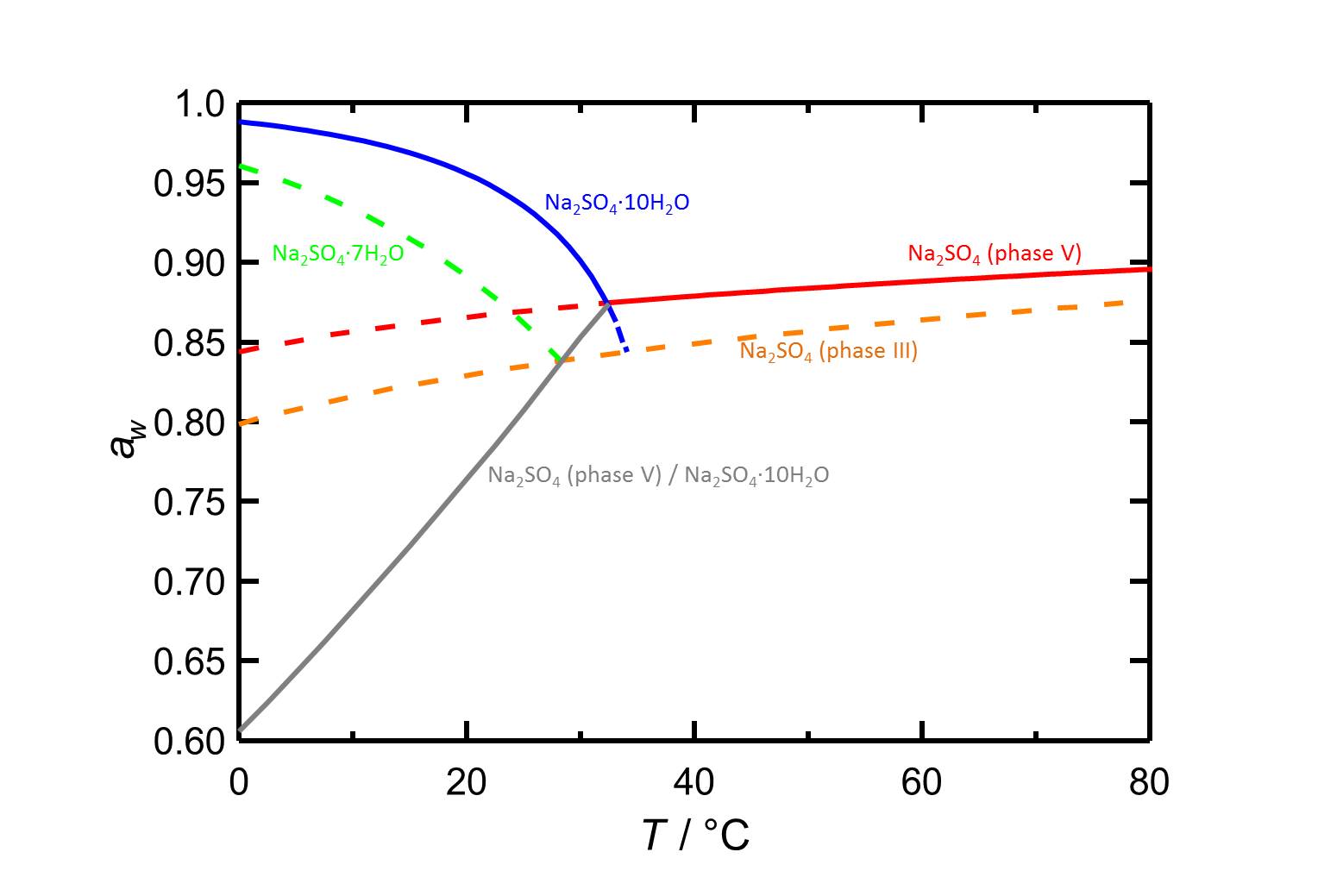
Phase Equilibrium of the Ternary System Na2SO3–Na2SO4–H2O at 293.15, 313.15, and 353.15 K | Journal of Chemical & Engineering Data
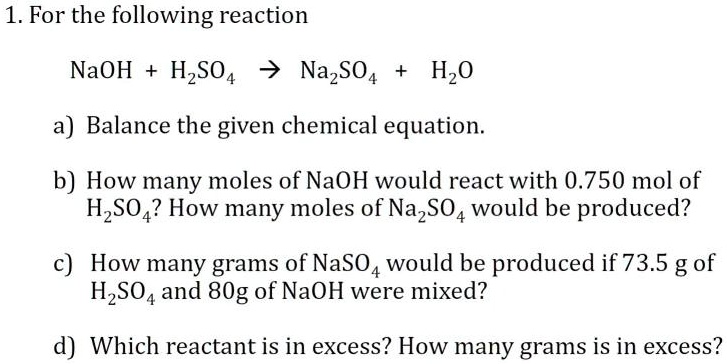
SOLVED: Texts: 1. For the following reaction: NaOH + HSO4 -> NaSO4 + H2O a) Balance the given chemical equation. b) How many moles of NaOH would react with 0.750 mol of

SOLVED: Which reaction represents an acid-base neutralization reaction? Hint: How do you identify acids and bases? O 2 NaOH + MgSO4 NaSO4 + Mg(OH)2 O HNO + KOH KNO3 + H2O O

OneClass: g. Using the following equation 2 NaOH + H2SO4 â†' 2 H2O + Naso, How many grams of sodium s...
![Sodium sulfate decahydrate [Na2SO4.10H2O] (Glauber's salt) Molecular Weight Calculation - Laboratory Notes Sodium sulfate decahydrate [Na2SO4.10H2O] (Glauber's salt) Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2022/11/sodium-sulfate-decahydrate-molecular-weight-calculation-300x191.jpg)
Sodium sulfate decahydrate [Na2SO4.10H2O] (Glauber's salt) Molecular Weight Calculation - Laboratory Notes

Jual NATRIUM SULFAT ANHIDRAT ( NaSO4.H2O ) per 50 gr Pro Analisa Merck Best Quality | Shopee Indonesia
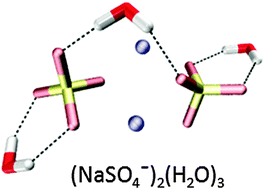
Probing the microsolvation of a quaternary ion complex: gas phase vibrational spectroscopy of (NaSO4−)2(H2O)n=0–6, 8 - Physical Chemistry Chemical Physics (RSC Publishing)

Using this balanced equation: 2 NaOH + H2SO4 —> H2O + Na2SO4 How many grams of sodium sulfate will be - brainly.com