Effect of Na2O/SiO2 and K2O/SiO2 mass ratios on the compressive strength of non-silicate metakaolin geopolymeric mortars - IOPscience

Membrane Crystallization of Sodium Carbonate for Carbon Dioxide Recovery: Effect of Impurities on the Crystal Morphology | Crystal Growth & Design

Water content diagram of the quaternary system K + // H 2 PO 4 2− , SO... | Download Scientific Diagram

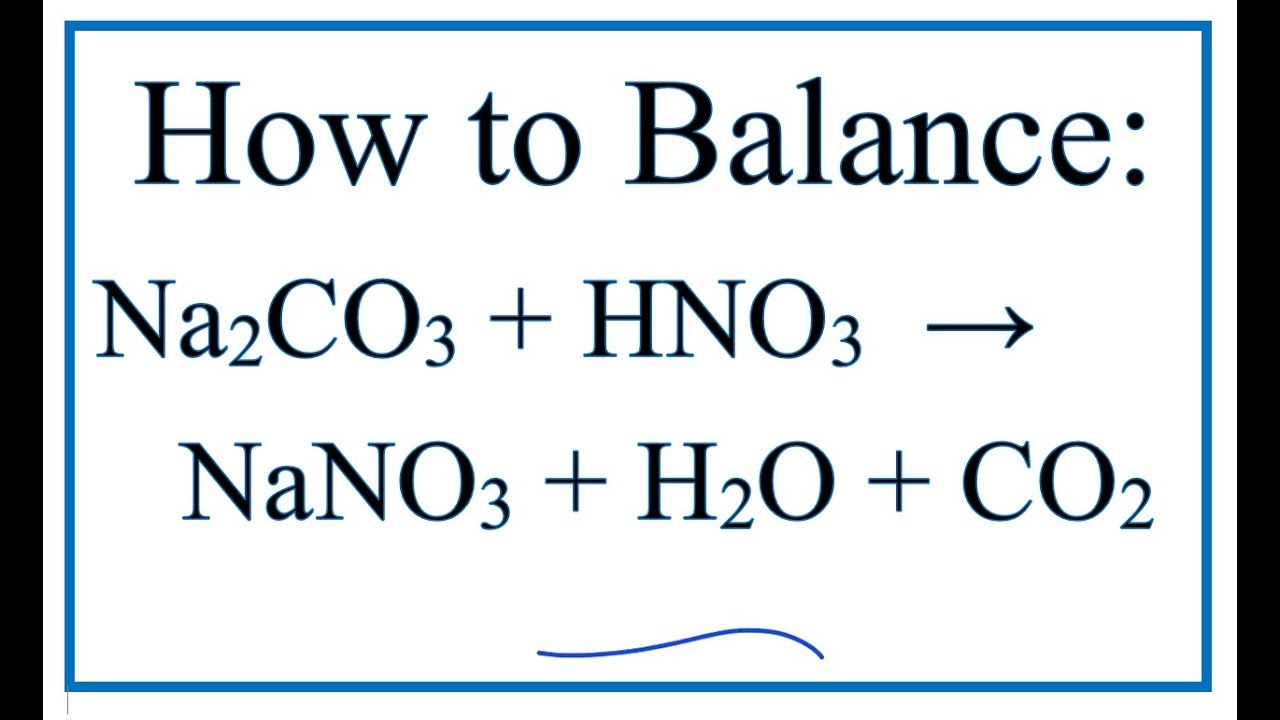
Na2CO3+HCl=NaCl+CO2+H2O Balanced Equation||Sodium carbonate+Hydrochloric acid=Sodium chloride+Carbon - YouTube

Sodium Carbonate (Na2CO3) Reaction: What does Sodium Carbonate do in a Reaction? - Aakash BYJU'S Blog

1 Dilute hydrochloric acid reacts with sodium carbonate solution. 2HCl(aq)+ Na2CO2(aq) - brainly.com

How to Balance Na2CO3+H2O→NaOH+CO2 | How to Balance Na2CO3+H2O→NaOH+CO2 | By Chemistry 360 | Facebook
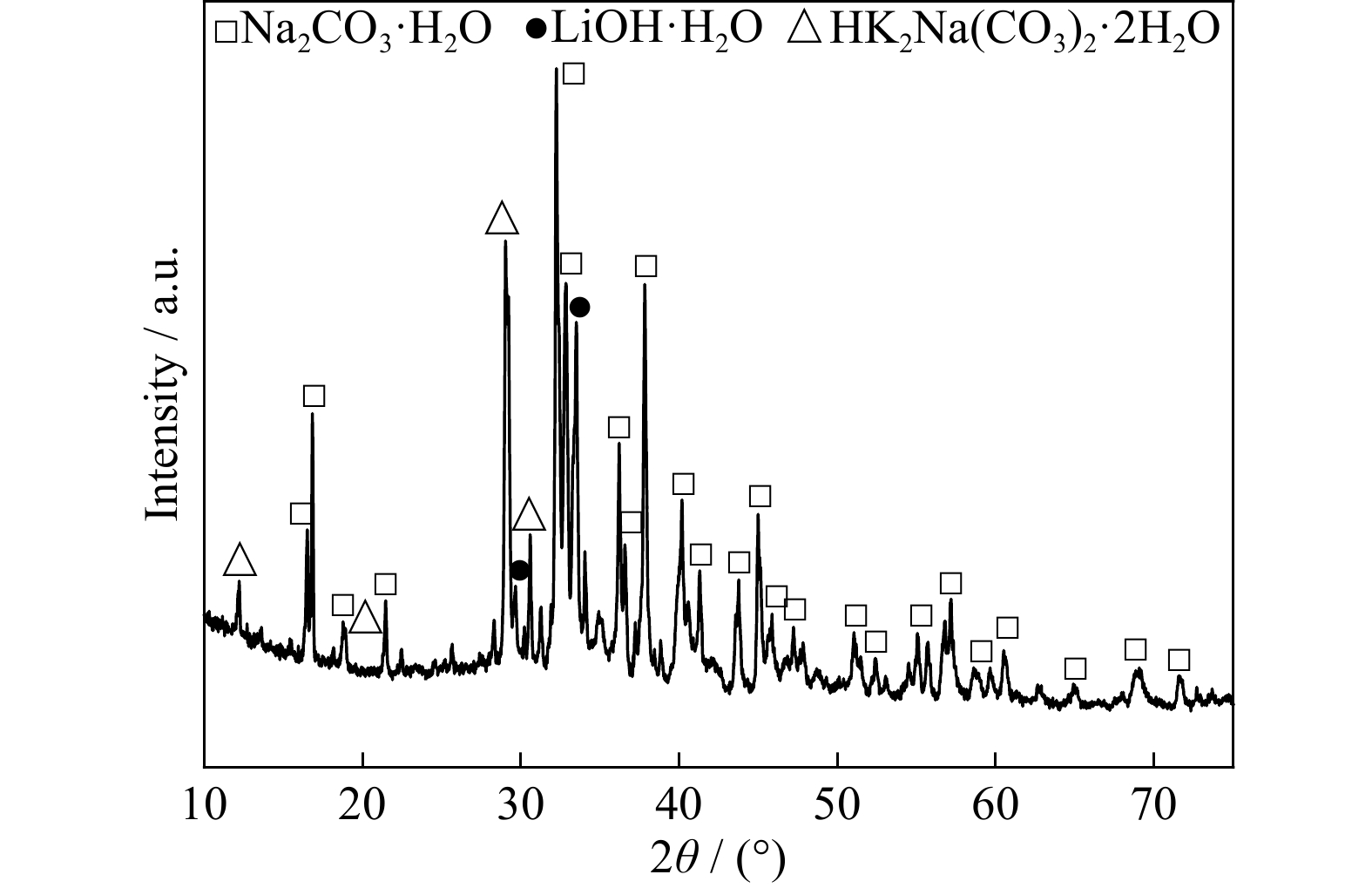
Extraction of lithium from the simulated pyrometallurgical slag of spent lithium-ion batteries by binary eutectic molten carbonates
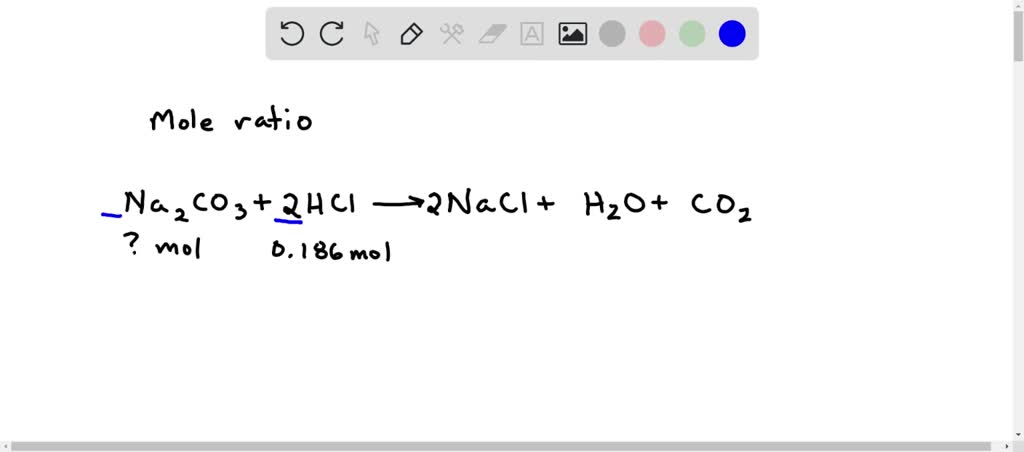
SOLVED: calculate the number of moles of sodium carbonate (Na2CO2) needed to neutralize 0,186 mole of HCI. chemical reaction : Na2CO3 + HCI —> NaCl + H20 + CO2
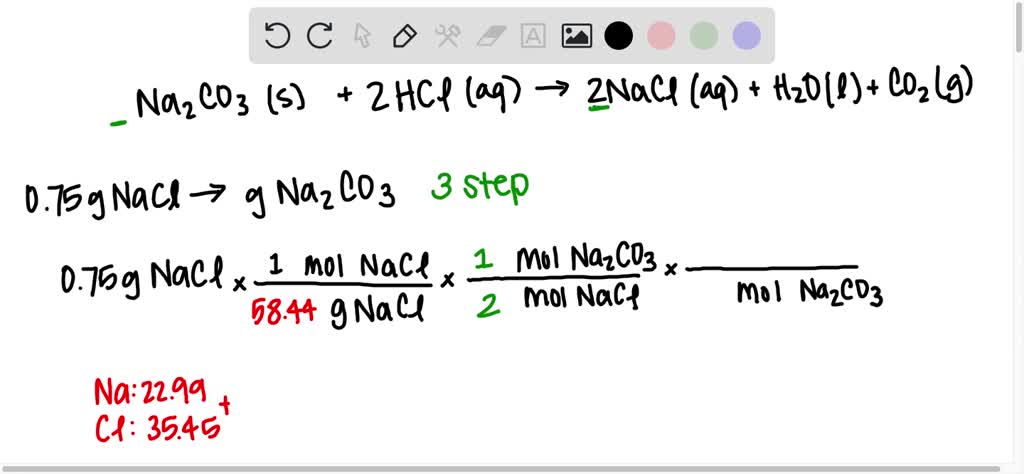
SOLVED: Na2CO3(s) + 2HCl(aq) â†' 2NaCl(aq) + H2O(l) + CO2(g) Using the balanced chemical reaction from step 1, calculate the mass of sodium carbonate (Na2CO3) that would be required to make 0.75

28. State as to why: (a) Aqueous solution of Na,co, is alkaline. (b) BaO is soluble but Baso, is insoluble in water. (1) Draw structure of BeClz (vapour). (m) Complete the following:

Ответы Mail.ru: Химия 8 класс! NaOH+CO=Na2CO2+H2O правильная ли это реакция и еще как мне получить Na2CO2?







![Odia] In the reaction, H2O2 +Na2CO3 rarrNa2O2 +CO2 +H2O,the substan Odia] In the reaction, H2O2 +Na2CO3 rarrNa2O2 +CO2 +H2O,the substan](https://static.doubtnut.com/ss/web-overlay-thumb/8303055.webp)