
KH2PO4 crystallisation from potassium chloride and ammonium dihydrogen phosphate – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on CyberLeninka open science hub.
![SOLVED: Describe stepwise how you would prepare 0.5 L of a 0.15 M phosphate buffer, pH 12.5, using KPO4·H2O (f.w. 230.28) and KH2PO4 (f.w. 174.18). [A-] pH = pKa + log [HA] SOLVED: Describe stepwise how you would prepare 0.5 L of a 0.15 M phosphate buffer, pH 12.5, using KPO4·H2O (f.w. 230.28) and KH2PO4 (f.w. 174.18). [A-] pH = pKa + log [HA]](https://cdn.numerade.com/ask_images/84c1fd22ef3046d0a647ee2adee981f8.jpg)
SOLVED: Describe stepwise how you would prepare 0.5 L of a 0.15 M phosphate buffer, pH 12.5, using KPO4·H2O (f.w. 230.28) and KH2PO4 (f.w. 174.18). [A-] pH = pKa + log [HA]
Comment calculer le pH d'une solution de KHCO3 (2M) et acide citrique (1M) (1:1) en présence d'un tampon phosphate (PBS : 10mM Na2HPO4 et KH2PO4 1,8 mM) ? - Quora

OneClass: Write the chemical reaction for:KH2PO4/K2HPO4 buffer solution + NaOH (aq)andWrite the chemi...

Thermodynamic Properties Data of Ternary System KBr–KH2PO4–H2O at 298.15 K | Journal of Chemical & Engineering Data

OneClass: Spectrophotometric analysis of phosphate can be performed by the following procedure: A. KH...

Calculate the pH of a buffer solution obtained by dissolving 25.0 g of KH2PO4(s) and 38.0 g of Na2HPO4(s) in water and then diluting to 1.00 L. | Homework.Study.com

SOLVED: A 0.492-g sample of KH2PO4 is titrated with 0.112 M NaOH, requiring 25.6 mL: H2PO4- + OH- → HPO42- + H2O What is the percent purity of the KH2PO4 (FW = 136.09)?
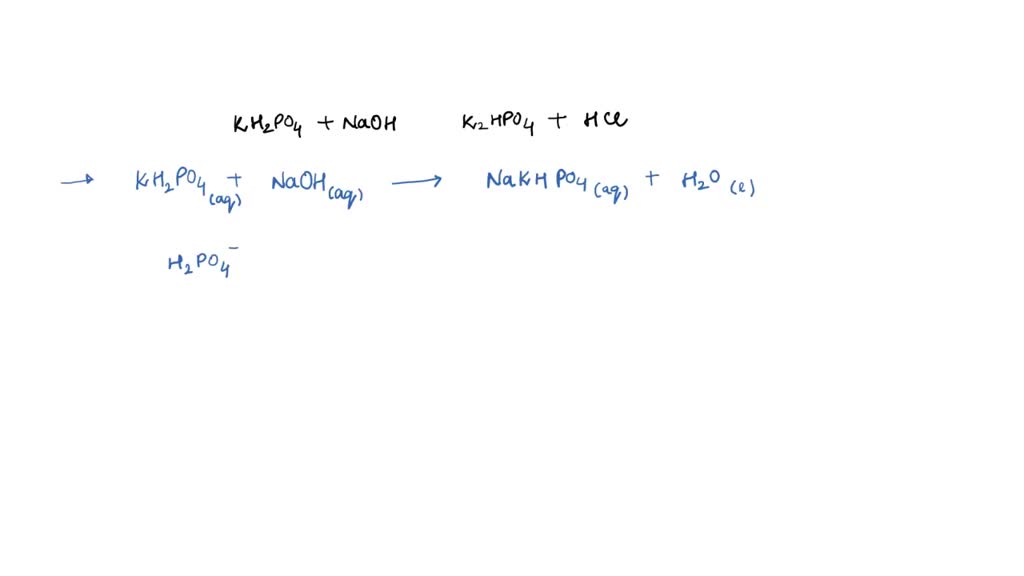
SOLVED: Write the equation and the reaction of the buffer solution KH2PO4 /K2HPO4 when NaOH and HCl is added
![FilSciHub Ed - CHEMISTRY MODULE] CHEMICAL REACTIONS & CHEMICAL EQUATIONS [ANSWER KEY] — Filipino Science Hub FilSciHub Ed - CHEMISTRY MODULE] CHEMICAL REACTIONS & CHEMICAL EQUATIONS [ANSWER KEY] — Filipino Science Hub](https://images.squarespace-cdn.com/content/v1/5f02d28f35d64d2a5022eeb1/1611724367429-9L5VAR6883R3CB69ME9Q/20.png)
FilSciHub Ed - CHEMISTRY MODULE] CHEMICAL REACTIONS & CHEMICAL EQUATIONS [ANSWER KEY] — Filipino Science Hub

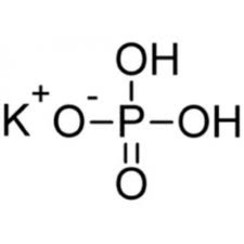

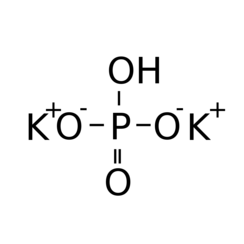

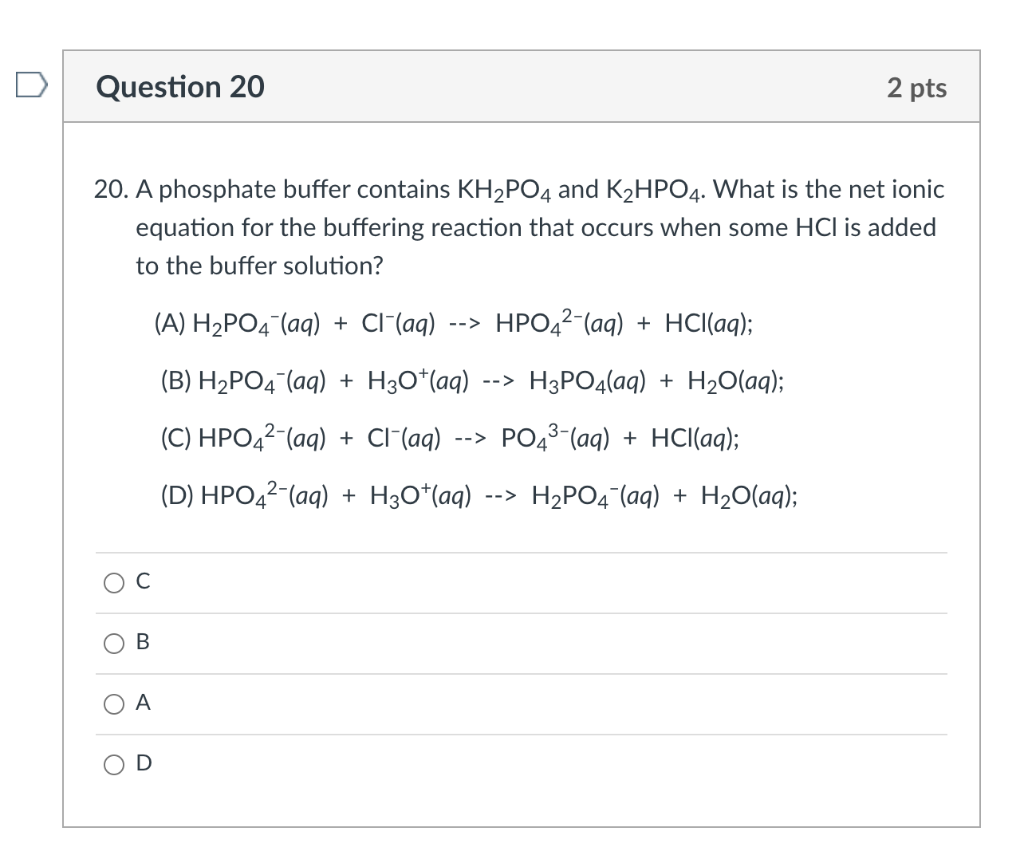





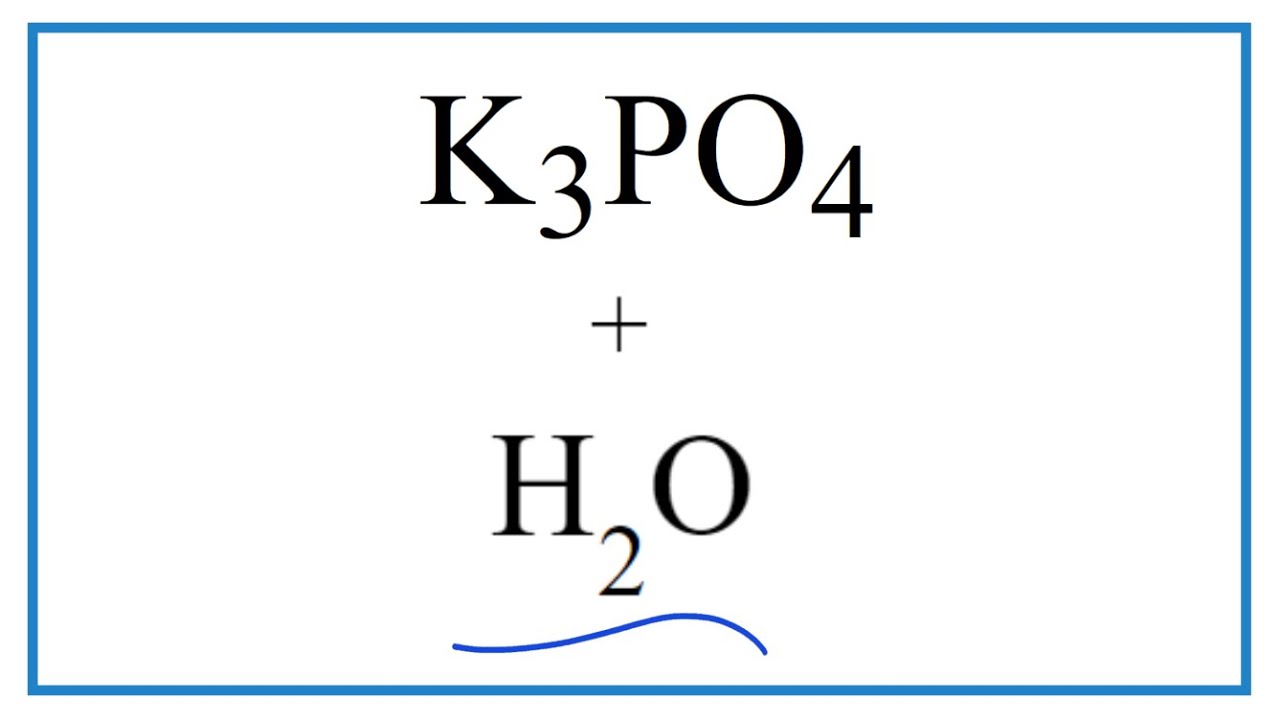
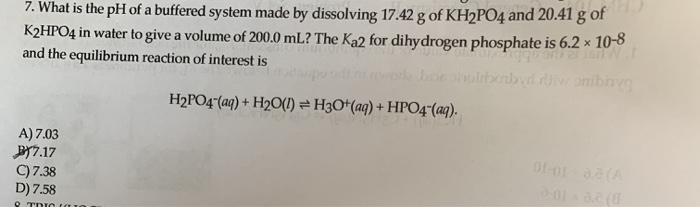
![ANSWERED] c 3 136 A 0 5 g sample of KH PO4 is titra... - Physical Chemistry - Kunduz ANSWERED] c 3 136 A 0 5 g sample of KH PO4 is titra... - Physical Chemistry - Kunduz](https://media.kunduz.com/media/sug-question-candidate/20200703122613132777-1655732.jpg)